Preparation of C20 Methallyl Rapamycin MaRAP
Gerald R. Crabtree
August 12th 1997
C20 Methally Rapamycin is a generally useful non toxic synthetic ligand or
CID (Chemical Inducer of Dimerization) that can be used to heterodimerize or
homodimerize proteins using classic FKBP12 and a mutant of the 89 aa FRB domain
of the human FRAP protein cloned by Stuart Scbreiber’s group. Ma rap is completely non toxic in yeast
as judged by the fact that none of the 6400 genes of yeast are reproducibly
altered in their level of expression by 6 hours incubation with 100 nM
drug. The drug also seems to be
non-toxic in animals and mouse embryos and is being used by the Crabtree
Laboratory for the production of strains of mice in which genes or their protein
products can be rapidly and reversibly inactivated or activated. The extinction
coefficient of C20 Marap is 12,200
at 238.
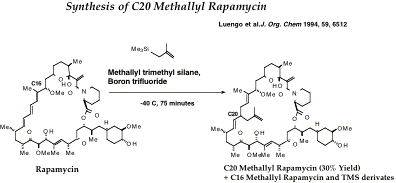
Reagents and Materials
1) Reaction vessel: 10 ml flask with small 1 cm stir bar and a rubber stopper that can be penetrated with needles. Place in heater at 80˚C and then desiccator prior to reaction.
2) Methallyl Silane: F.W. 119. Prepared from methallylsilane (See below).
3) Boron triflouride etherate: (C2H6)2O.BF3 Aldrich 21,660-7 F.W. 141.93.
4) Rapamycin: 25 milligrams stored dry under Dryrite at -20˚C.
5) ANHYDROUS Methylene chloride 3 ml. Recently distilled.
6) Dry ice acetylnitrile bath in a 200 ml insulted container, check to see that it is about -40 ˚C.
7) Glass syringes One 3 ml with long flexible needle.
Two 100 ul syringes with moderately long flexible needle.
8) Saturated NaHCO3.
9) Brine. Saturated NaCl.
10) Desiccator with Dryite and vacuum outlet.
11) TLC plates- silica gel 60F254 from EM Separation Technology #5717-7 20X20.
12) TLC solution- 7.5 ml Ethyl Acetate, 2.5 ml Hexane, 100 ml of ethanol.
13) Titan™ polyproplyene 1 micron filters, HPLC grade with 0 UV absorbance.
14) Thermometer capable of measuring temperatures to -40˚C.
15) Rotovac.
16) Flask for rotovac preweighed.
17) UV Light.
18) LC-908 Recycling HPLC.
Procedure
Heterlytic
Cleavage at C16 and SN1 Addition of Methallyl
1) Weigh out the rapamycin (1 equivalent) into dry 10 ml glass flask with the stirring bar in place.
The flask should come directly from the desiccator as should the stir bar.
Rap weight______. (aim for about 25 mg).
2) Immediately purge the flask with dry nitrogen and maintain under positive nitrogen pressure just over atmospheric pressure.
3) Place the -40˚C bath on a magnetic stirrer and adjust until it is just below the flask.
4) Using the 3 ml syringe with the long needle remove 3.5 ml of CH2Cl2 from the distillation apparatus. Immediately reseal the distillation apparatus.
5) Add immediately to the reaction flask and turn on the stirrer to dissolve the rapamycin. Visually carefully check to see that the rapamycin is dissolved.
6) After the rap is dissolved lower the flask into the -40˚C bath leaving enough visible to see the tip of a syringe needle inserted through the rubber stopper.
7) Add 50 ml (10 equivalents) of the methallyl silane to the rap solution.
volume added___________Time_____
8) Continue stirring until all the reagents are completely combined (about 1 minute). Be very careful that there is no reagent on the side of the flask that is not combined since the next step will convert any rapamycin to the carbocation.
9) Make up a solution of BF3 etherate in benzene by adding 100 ul of BF3 to 400 of benzene. Add 200 ml (10 equivalents) of the BF3 solution to the flask by carefully dripping it into the flask one drop at a time.
volume added___________Time______. Continue the incubation, adding dry ice occasionally to the ice bath to maintain it near -40˚C for 75 minutes. The solution will turn as brown as a Jersey Cow and then become nearly clear, slightly yellow.
Collection
and Analysis of Reaction Products
10) At the end of the reaction transfer the contents of the vial to a flask containing 3 ml of saturated NaCHO3, by using the pressure provided by the nitrogen to transfer the reaction through a stainless steel flexible tube to the flask containing the saturated NaHCO3, which should be stirring vigorously during the canulation. This tube must also be completely dry and freshly taken out of the desiccator. Once the reaction mixture is transferred to the NaHCO3 the acid is neutralized and the reaction can not proceed.
11) Clean the reaction vial of the remainder of the reactants by washing the walls with anhydrous methylene chloride and transferring it to the saturated NaHCO3 solution through the canulation tube.
12) After stirring the combination of the reaction mixture and the saturated NaHCO3 for about 10 minutes, add 5 ml of ethyl acetate and continue stirring for 1 to 2 minutes. Let set till the two layers separate.
13) Remove the bottom, aqueous layer and add an equal volume of brine to the organic layer to remove water by osmosis.
14) Remove the lower brine layer. The interface here is very hard to see and requires that you place your eye parallel to the layers being separated.
15) Add about 1 gram of anhydrous Sodium Sulfate (Fisher 5421-500) to the organic layer. This should remove any residue water. If much water remains this will result in a warming of the solution.
16) Dot about 1 ml on to a TLC plate along with a Ma Rap standard and a rapamycin standard. The Ma Rap migrates abherrently in ethanol and thus it must be dried down and resuspended in methylene chloride to run with the newly synthesized material.
17) If there has been substantial conversion to Ma Rap, the solution should be filtered first through a cotton pipett filter and then through a Titan™ 1 micron filter (UV absorbance 0, HPLC grade).
18) Collect the filtered solution into a preweighed roto vac flask.
19) Roto vac under laboratory vacuum until dry. Store sealed for no longer than overnight.
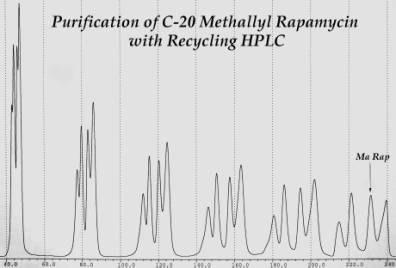
Separation
of the Methallyl Rapamycin Products by Recycling HPLC.
The products of the reaction will be a mixture of the R- and S- diasteromers and several other less well characterized stuff. These chiral products must be resolved since the S form is able to bind to FRP and produce rapamycin toxicity. The column that is used for this is polystyrene. The LC-908 HPLC allows one to make multiple passes through the same column to optimize separation and eliminate unwanted peaks.
1) Turn on the LC-908 Recycling HPLC with the “Main Power” Switch.
2) Check the chloroform supply. The bottle should be about 1/2 full.
3) Connect the chart recorder and the MacIntosh SMAD interface if you want to analyze the peaks and recovery. Turn on the button on the back of the SMAD interface.
4) Purge the line of any accumulated bubbles as described in the manual.
5) Place in collect and syphon mode and collect chloroform to a waste bottle for about 60 minutes to be certain that any residual from the last purification will not contaminate your sample. The flow rate is about 3 ml per minute and the pressure should be between 30 and 45 kg/cm2. Return to the recycle mode.
6) While this is happening load the reaction mixture into a 1 ml syringe from the roto-vac flask.
7) Clean the syringe (1 ml glass) by 3 washes with chloroform.
8) Clean the injector port by injecting 3 times with chloroform. Collect the waste from the container in the middle window of the HPLC.
Injection
9) Dissolve the reaction products up in 100 ul of chloroform, place in a 250 ul syringe and place the HPLC injection port in the “Load” mode. Insert the syringe and with the syringe still in place turn the switch to “Inject”. There will be a loud noise as the chloroform inters the chamber.
10 Record the time of injection to the second ___________.
11) The R rap peak has a retention time of about 33 minutes and 38 seconds. The typical profile will have a slightly separated R and S peak and perhaps 2 more peaks. Peak A is a silated form of R and is biologically active on the mutant, Peak B is the S form, Peak C is the R form and Peak D is another silated form, probably the S.
12) Let the machine cycle for about 40 minutes and examine the profile.
13) If possible, make a judgment about the peaks to cut and do the first cut on the second or third pass.
14) To cut a peak, place the collection tubing into a tube and switch to “collect”.
15) To clean up the machine, past chloroform to waste for about 1 hour. Turn off the chart recorders and the SMAD interface and save the Mac file.
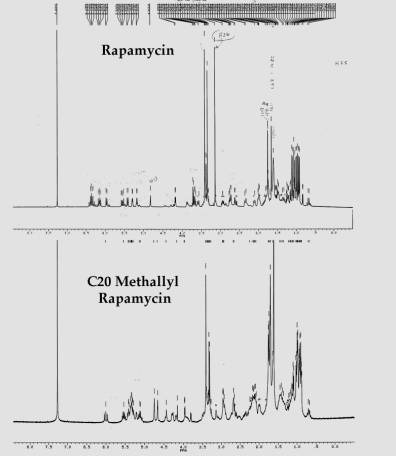
Characterization of methallyl
rapamycin.
The molecular weight should be 960/937 by mass spect.
The UV spectum of the C16 S from is very much like rapamycin with a max at 280 and two smaller peaks on either side. The C20 is completely different and should have a peak at 238 to 242. The extinction coefficient of C20 Marap is 12,200 at 238.
The NMR pattern should show the following diagnostic patterns:
1) Loss of the Methoxy peak at 3.1 ppm.
2) Addition of the methyl peak at 1.6 ppm.
3) Addition of the vinyl carbon peak at 4.65 ppm.
4) The R form should show loss of the peaks greater than 6 ppm and a radical. change in the olifin protons from 5.0 to 5.65 from both rapamycin and the S form.
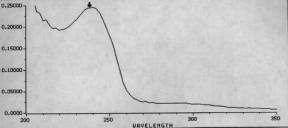
UV spectrum. The easiest way to tell if you have the right thing is to just do a uv scan. If you inject rapamycin you will note a loss of the triene peaks and a single peak for the C20. This of course results from the loss of the triene structure characteristic of rapamycin.The marpa peak should be at about 237.
Preparation of Methallyl
Silane
Reference: Hagen, G., Mayr, H. JACS 1991, 113, 4954
3-Chloro 2 methyl propene (Aldrich 10803-0) FW 90.55 3.12 gm or 2 eq.
Chlorotrimethylsilane (Ald 38652-9) FW 108.64-- 1.87 gm or 1 eq.
Magnesium 1.14 gm FW 24 1.14 gm or 2.75 eq.
3-Chloro 2 methyl propene in 35 ml THF added dropwise to Chlorotrimethylsilane in 20 ml THF to magnesium turnings under THF.
Initiate reaction by heating to about 60˚C with caution. When reaction intitates as detected by the dissolving of the Mg, maintain at 0 C.
Heated to 65˚C for 4.5 hours.
Stirred overnight.
added 1:1 NH4/NH4Cl saturated.
Decanted into a separatory funnel.
Placed on Cold bath roto vac at 500-600 torr.
Distillation and Fractionation as follows.
F1 THF 61-65˚C
F2 65 - 70˚C
F3 70-75˚C
F4 85-100˚C
Note! dimers of methyl propene may contaminate the product, particularly in the lower temperature fractions.